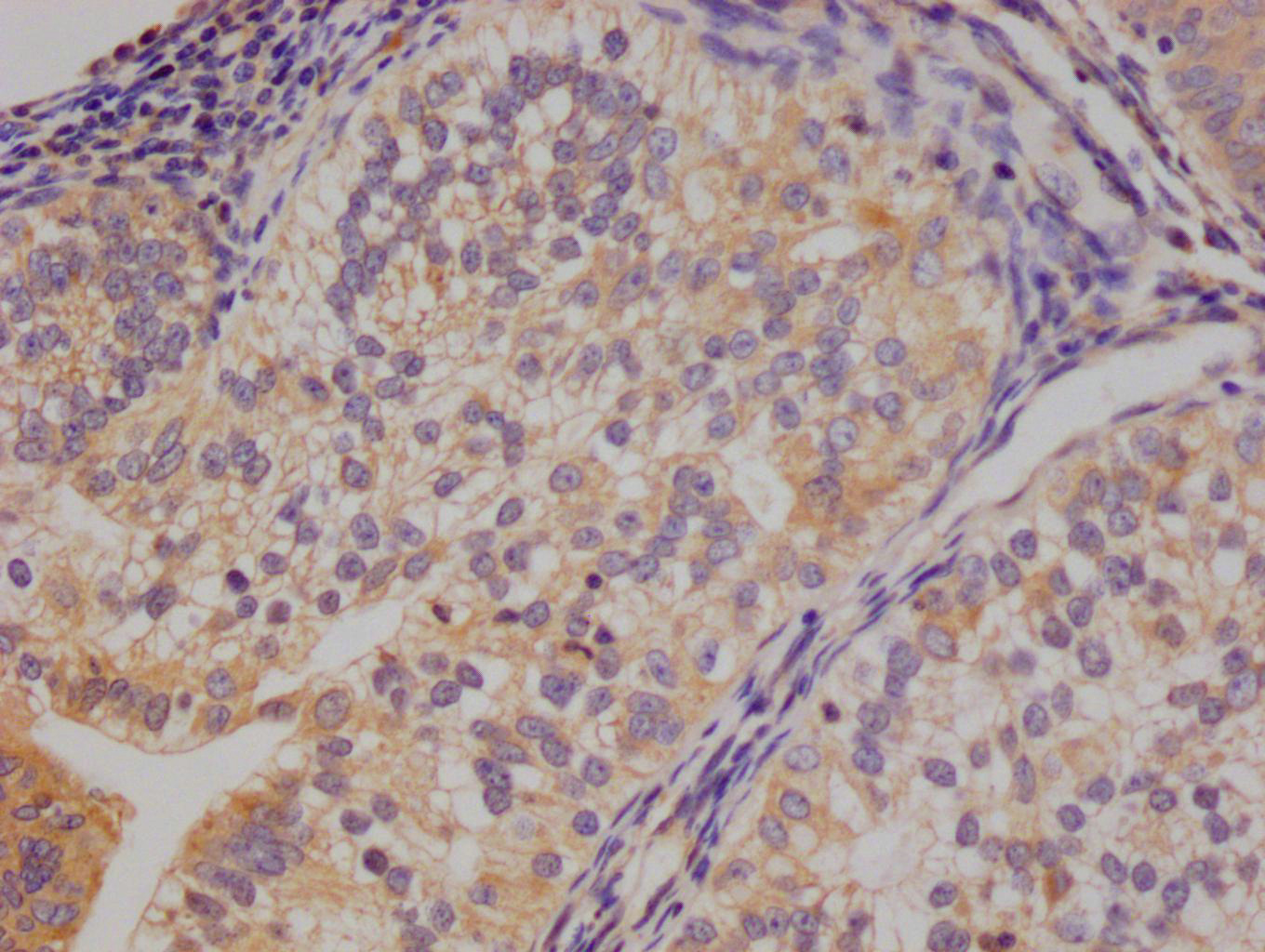
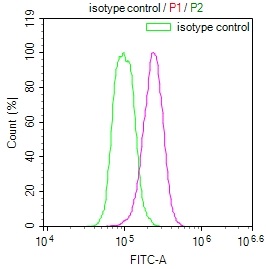
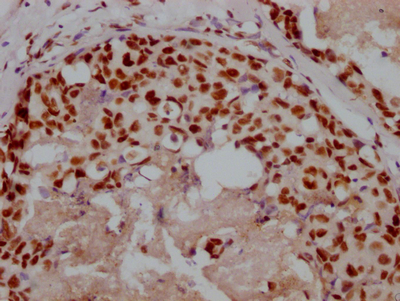
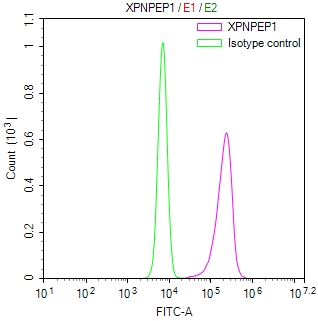
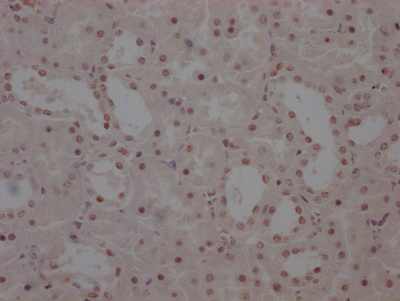
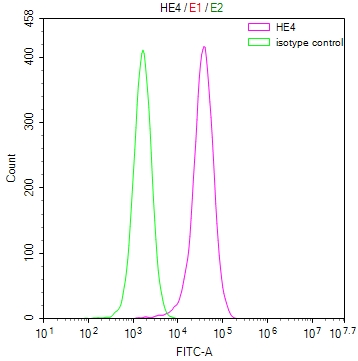
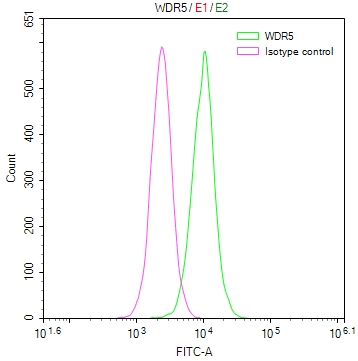
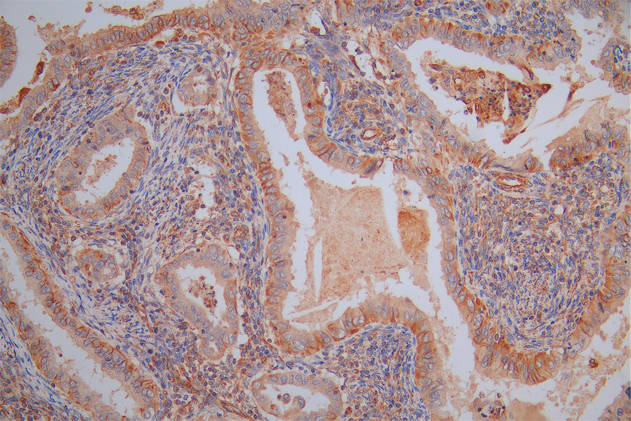
IAPP Antibody, FITC conjugated
-
中文名稱:IAPP兔多克隆抗體, FITC偶聯
-
貨號:CSB-PA12224C0Rb
-
規格:¥880
-
其他:
產品詳情
-
產品名稱:Rabbit anti-Homo sapiens (Human) IAPP Polyclonal antibody
-
Uniprot No.:
-
基因名:
-
別名:Amylin antibody; DAP antibody; Diabetes associated peptide antibody; Diabetes-associated peptide antibody; IAP antibody; IAPP antibody; IAPP_HUMAN antibody; Insulinoma amyloid peptide antibody; Islet amyloid polypeptide (diabetes associated peptide, amylin) antibody; Islet amyloid polypeptide antibody
-
宿主:Rabbit
-
反應種屬:Human
-
免疫原:Recombinant Human Islet amyloid polypeptide protein (34-70AA)
-
免疫原種屬:Homo sapiens (Human)
-
標記方式:FITC
-
克隆類型:Polyclonal
-
抗體亞型:IgG
-
純化方式:>95%, Protein G purified
-
濃度:It differs from different batches. Please contact us to confirm it.
-
保存緩沖液:Preservative: 0.03% Proclin 300
Constituents: 50% Glycerol, 0.01M PBS, PH 7.4 -
產品提供形式:Liquid
-
儲存條件:Upon receipt, store at -20°C or -80°C. Avoid repeated freeze.
-
貨期:Basically, we can dispatch the products out in 1-3 working days after receiving your orders. Delivery time maybe differs from different purchasing way or location, please kindly consult your local distributors for specific delivery time.
-
用途:For Research Use Only. Not for use in diagnostic or therapeutic procedures.
相關產品
靶點詳情
-
功能:Selectively inhibits insulin-stimulated glucose utilization and glycogen deposition in muscle, while not affecting adipocyte glucose metabolism.
-
基因功能參考文獻:
- In conclusion, IAPP/amylin directly interacts with NLRP3 to activate NLRP3 inflammasome, and this interaction could be an attractive drug target to avoid inflammation and beta-cell death during therapy for diabetes, although there are mechanisms in NLRP3 inflammasome and diabetes pathology in human tissues that still require elucidation. PMID: 30014749
- These findings suggest that increased membrane permeation induced by oligomeratization of amylin peptide in cell sarcolemma contributes to Ca(2+) dysregulation in pre-diabetes. PMID: 29604965
- down-regulation of IAPP expression induces the death of human annulus fibrosus cells via mitochondrial and death receptor pathways, potentially offering a novel therapeutic target for the treatment of intervertebral disc degeneration. PMID: 28433710
- This study highlights that with positional scanning of the split-tetracysteine motif (Cys-Cys), the fluorogenic probe fluorescein arsenical hairpin detection method offers unique time-dependent conformational insights on the proteospecies assembled throughout the amyloidogenic pathway of IAPP. PMID: 29360346
- Bri2 BRICHOS domain is a potent inhibitor of IAPP fibril formation.IAPP colocalizes with Bri2 both intracellularly and in islet amyloid deposits. PMID: 29507232
- Insulin resistance in rheumatoid arthritis does not appear to be mediated by amylin. This would imply that the mechanisms associated with IR in RA patients differ from those at work in type 2 diabetes. PMID: 29352842
- During aggregation, the nucleating NFGAIL region remains flexible and accessible within this isolated intermediate, suggesting a mechanism by which membrane-associated aggregation may be propagated. PMID: 29148426
- Study presents a new Zn(2+) binding site in the N-terminus of fibrillary amylin with three different coordination modes. Simulations showed that Zn(2+) ions bind to polymorphic amylin fibrils with a preference to bind to four Cys residues rather than two Cys residues of two neighboring amylin monomers. PMID: 28692245
- The IAPP beta-hairpin can serve as a molecular recognition motif enabling control of IAPP aggregation. PMID: 27641459
- cholesterol significantly modulates the ability of model membranes to induce IAPP amyloid formation and IAPP-mediated membrane damage. PMID: 29373018
- point mutations within the central aggregation-prone regions contribute to the reduction of the overall amyloidogenic potential of IAPP but do not completely abolish the formation of IAPP amyloid fibrils. PMID: 28602716
- Using the Tg2576 AD mouse model, a single intraperitoneal injection of amylin significantly increased Abeta serum levels, and the effect was abolished by AC253, an amylin receptor antagonist, suggesting that amylin effect could be mediated by its receptor. Subsequent mechanistic studies showed amylin enhanced Abeta transport across a cell-based model of the blood-brain barrier. PMID: 28059785
- These results suggest that protein segment structures represent polymorphs of their parent protein and that segment 19-29 S20G may serve as a model for the toxic spine of human IAPP. PMID: 28045370
- All-atom explicit-water molecular dynamics (MD) simulations studying adsorption, orientation, and surface interaction of hIAPP aggregates with different sizes (monomer to tetramer) and conformations (monomer with alpha-helix and tetramer with beta-sheet-rich U-turn) upon adsorption. hIAPP monomer with alpha-helical conformation and hIAPP pentamer with beta-sheet conformation can adsorb on both POPC and POPC/POPE bilayers. PMID: 28585804
- The aggregation of rhIAPP also occurred significantly faster when compared with that of the chemically synthesized peptide. PMID: 29046394
- Data (including data from studies using tissues from transgenic mice) suggest that IL1B plays dual roles by (1) mediating islet amyloid-induced FAS up-regulation and apoptosis in pancreatic beta-cells and (2) down-regulating IAPP precursor processing thereby potentiating islet amyloid formation. (IL1B = interleukin-1beta; FAS = FAS cell surface death receptor; IAPP = islet amyloid polypeptide) PMID: 28058779
- Data suggest that single aromatic/hydrophobic amino acid residues within IAPP (islet amyloid polypeptide) amyloid core region are able to control its interaction with amyloid-beta(1-40) or amyloid-beta(1-42) but not IAPP self-assembly; four aromatic/hydrophobic residues are able to control both IAPP amyloid self-assembly and its cross-interaction with amyloid-beta(1-40) or amyloid-beta(1-42). PMID: 28684415
- Data show that aluminum (Al3+) could inhibit islet amyloid polypeptide hIAPP(11-28) fibrillogenesis. PMID: 28338927
- The absence of BACE2 ameliorates glucose tolerance defects induced by IAPP overexpression in the beta-cell and promotes beta-cell survival. PMID: 28337562
- This study supports the elucidation of the structural basis of IAPP amyloid formation and highlights the extent of amyloid fibril polymorphism. PMID: 27607147
- Data suggest that a single GlcNAc residue at CTR N130 (asparagine 130) is responsible for enhanced affinity of calcitonin for CTR ECD; the same appears to apply for enhanced affinity of amylin for RAMP2-CTR ECD. [GlcNAc = N-acetylglucosamine; CTR = calcitonin receptor; ECD = extracellular domain; RAMP2 = receptor (calcitonin) activity modifying protein 2]. PMID: 28614667
- The kinetics of human amylin amyloid formation can be monitored by SYPRO-orange fluorescence and match the time course determined with thioflavin-T assays. PMID: 27479186
- effect of cholesterol on the amyloidogenicity of IAPP PMID: 27410742
- Together, our preclinical results suggest that AT406 could be further evaluated as a promising anti-pancreatic cancer agent. PMID: 27387230
- Al(III) could promote fibrillation and aggregation of hIAPP, while EGCG could inhibit the fibrillation of hIAPP and lead to the formation of hIAPP amorphous aggregates instead of the ordered fibrils PMID: 28074190
- Chondroitin sulfate A has an intensive promotion effect on the fibrillation of human IAPP at the palmitoyloleoylphosphatidylcholine (POPC) membrane, which is larger than the total effect of Chondroitin sulfate A alone and POPC alone. PMID: 27067251
- C4BP protects beta-cells from IAPP cytotoxicity by modulating IAPP fibril formation extracellularly and also, after uptake by the cells, by enhancing cholesterol synthesis. PMID: 27566545
- beta-hairpin peptide inhibitor of IAPP aggregation that are stabilized in that conformation, or even forced to remain in the hairpin conformation by a backbone cyclization constraint, display superior activity as inhibitors. PMID: 27317951
- hA17-29 aggregate toxicity seems to be mediated by RAGE and p75-NGFR receptors PMID: 27804051
- Serum levels of preptin and amylin were significantly lower in patients with psoriasis and Behcet's disease. PMID: 25545917
- These data suggest participation by both soluble and fibrillar aggregates in IAPP-induced islet inflammation. IAPP-induced activation of TLR2 and secretion of IL-1 may be important therapeutic targets to prevent amyloid-associated beta cell dysfunction. PMID: 26786104
- the effect of the endoplasmic reticulum chaperone protein disulfide isomerase (PDI) on beta-cell dysfunction, was examined. PMID: 26607804
- Data suggest that IAPP/membrane interaction strongly depends on protonation state of His18; at neutral pH, N-terminal domain is stabilized/anchored to membrane, while C-terminal domain is disordered as if in solution. PMID: 26953503
- Surface molecular structure and amino acid residue composition of hIAPP fibrils are specifically probed with nanoscale resolution using tip-enhanced Raman spectroscopy. PMID: 25952953
- Structural studies and cytotoxicity assays of "aggregation-prone" IAPP(8-16) and its non-amyloidogenic variants suggest its important role in fibrillogenesis and cytotoxicity of human amylin. PMID: 25913357
- structural and dynamical information of amylin and CGRP PMID: 26331261
- The computational models support the cross-sequence interactions between ABETA and IAPP pentamers, which would lead to the complex hybrid ABETA-IAPP assemblies. PMID: 26173078
- Study focused on human islet amyloid polypeptide aggregates and a de novo designed short polypeptide at lipid/water and air/glass interfaces; found that parallel beta-sheets adopt distinct orientations at various interfaces and exhibit characteristic chiroptical responses in the amide I and N-H stretch regions. PMID: 26263128
- Secondary Structure of Rat and Human Amylin PMID: 26221949
- The intramolecular hydrogen bond formation by His(18) and the possibility of His(18) participating in the formation of alpha-helical structures greatly modulated the manner of hIAPP amyloid formation. PMID: 26777153
- Matrix Metalloproteinase-9 Protects Islets from Amyloid-induced Toxicity. PMID: 26483547
- cross-seeding assemblies between hIAPP and rIAPP oligomers PMID: 25706385
- combined computational and experimental study offers detailed mechanistic insight into the complex role of zinc on IAPP aggregation and T2D development PMID: 26603575
- When copper(II) was present in the solution, no dimers were detected. Thus, the copper(II) ions disrupt the association pathway to the formation of beta-sheet rich amylin fibrils. PMID: 26352401
- Computational studies identified new IAPP mutations that have stronger amyloid fibrils destabilizing potential that the currently known. PMID: 25903685
- The effect of a single proline mutant at position 26 in an amylin polypeptide on its binding to a lipid bilayer was studied. PMID: 25427619
- Inhibition of IAPP aggregation by insulin depends on the insulin oligomeric state regulated by zinc ion concentration PMID: 25649462
- Introducing the porcine insulin promoter-hIAPP expression vector into PK15 cells combined with exogenous Pdx-1, MafA and NeuroD1 resulted in the efficient expression of hIAPP at the gene level, but not the protein. PMID: 24825179
- There was abundant amylin aggregates in lysates of cardiac myocytes from obese patients, but not in controls. amylin aggregation at the sarcolemma induces oxidative stress and Ca(2+) dysregulation. PMID: 25146704
- interaction between HS and IAPP or the subsequent effects represent a possible therapeutic target whose blockage can lead to a prolonged survival of beta cells PMID: 25922077
顯示更多
收起更多
-
亞細胞定位:Secreted.
-
蛋白家族:Calcitonin family
-
數據庫鏈接:
Most popular with customers
-
-
YWHAB Recombinant Monoclonal Antibody
Applications: ELISA, WB, IHC, IF, FC
Species Reactivity: Human, Mouse, Rat
-
-
-
-
-
-